
A company that develops medical products approached us with an all too common problem. They were required to test their medical device to the new IEC 60601-1-2, 4th Edition standards to maintain their compliance certification. But the device was designed prior to the latest revision of the 4th Edition on electromagnetic compatibility (EMC), and so it ultimately failed the new, more stringent requirements for electrostatic discharge (ESD) and radiated immunity.
We worked closely with SILENT Solutions LLC, an expert EMC consulting company and a DornerWorks’ partner, to identify the root cause of the certification test failures. SILENT Solutions LLC determined that the printed circuit board (PCB) layout was not conducive to proper emissions or immunity standards to pass the updated regulations. They created a report of the recommended countermeasures and submitted it to the company. The company then reached out to us for our expert hardware consulting services to perform the needed PCB design updates to address the report’s findings.

Upon inspection of the PCB layout that contained a microprocessor, we found several areas needing improvement in the power distribution network (PDN). There was also an opportunity for applying some EMC/EMI best practices that had not been implemented. Working closely with SILENT Solutions LLC, we designed a new PCB layout that maintained the size and form factor of the customer’s current device and fit into the existing mechanical enclosure. This minimized the cost of the changes required to pass the new testing.
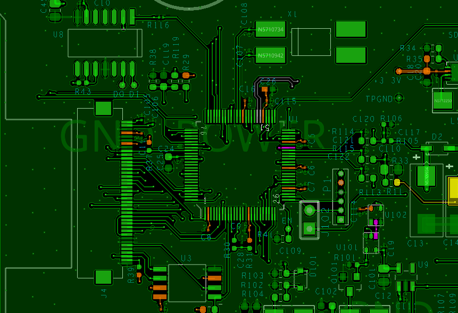
The DornerWorks design was built and tested for to verify functionality. Then the prototypes were sent to the customer for 4th Edition testing. The updated devices passed the testing of the revised standards on the first try and the customer was able to recertify the device by only updating the PCB. This further saved the customer money, as they could retain their product’s original mechanical housings and peripheral components (batteries, LCD displays, etc.) with zero modifications.
Thanks to the collaboration between DornerWorks and our partner, SILENT Solutions LLC, we were able to provide the customer with a quick, cost-effective solution to an increasingly common compliance problem. If this is a problem you’re facing today, we’d love to help you maintain your hardware compliance certification!
Schedule a meeting with us today to see how we can help you address your regulatory headaches.
Regulatory standards are changing. Don’t get forced into an early obsolescence.
Sign up for our development series on mitigating product risk associated with medical design standards updates and prepare your team to move forward with confidence.
Download the series